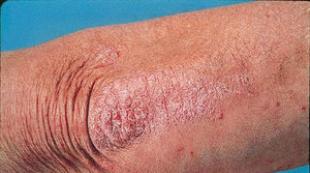
Benzylpenicillin sodium salt time to reach maximum concentration. Features of the effect of the drug on the ability to drive a vehicle or potentially dangerous mechanisms
Name
powder for the preparation of solution for injection and local application 500000 units, 1000000 units
Pharmacotherapeutic group
biosynthetic antibiotic penicillin
Trade name
Benzylpenicillin sodium salt
International nonproprietary name
benzylpenicillin
Dosage form
powder for the preparation of solution for injection and topical use.
Compound
Active substance: Benzylpenicillin sodium (benzylpenicillin sodium salt) - 500,000 units and 1,000,000 units.
ATX code
Pharmacological properties
Pharmacodynamics
Bactericidal antibiotic from the group of biosynthetic (“natural”) penicillins. Suppresses the synthesis of the cell wall of microorganisms. Active against gram-positive pathogens: Staphylococcus spp. (non-penicillinase-forming), Streptococcus spp. (including Streptococcus pneumoniae), Сorynebacterium spp. (including Corynebacterium diphtheriae), Bacillus anthracis, Actinomyces spp.; gram-negative microorganisms: Neisseria gonorrhoeae, Neisseria meningitidis, as well as against Treponema spp., class Spirochaetes. Not active against most gram-negative bacteria (including Pseudomonas aeruginosa), Rickettsia spp., viruses, protozoa. Staphylococcus spp., which produce penicillinase, are resistant to the drug.
Pharmacokinetics
The time required to achieve maximum concentration in blood plasma when administered intramuscularly is 20-30 minutes. Communication with plasma proteins - 60%. Penetrates organs, tissues and biological fluids, except for cerebrospinal fluid, eye tissue and prostate gland, with inflammation meningeal membranes penetrates the blood-brain barrier. It is excreted unchanged by the kidneys. The half-life is 30-60 minutes, with renal failure- 4-10 hours or more.
Indications for use
Bacterial infections caused by penicillin-sensitive pathogens: community-acquired pneumonia, pleural empyema, bronchitis; sepsis, septic endocarditis (acute and subacute), peritonitis; meningitis; osteomyelitis; infections genitourinary system(pyelonephritis, pyelitis, cystitis, urethritis, cervicitis), biliary tract (cholangitis, cholecystitis); wound infection, infections of the skin and soft tissues: erysipelas, impetigo, secondary infected dermatoses; diphtheria; scarlet fever; anthrax; actinomycosis; ENT infections, eye diseases ( acute conjunctivitis, corneal ulcer, etc.); gonorrhea, syphilis.
Contraindications
Hypersensitivity, including to other penicillins, cephalosporins.
Use during pregnancy
The use of the drug during pregnancy is possible only if the expected benefit to the mother outweighs the potential risk to the fetus.
If it is necessary to prescribe the drug during lactation, breastfeeding should be stopped.
Directions for use and doses
Benzylpenicillin and sodium salt are administered intramuscularly, intravenously, subcutaneously, locally.
Intravenously and intramuscularly:at average severe course diseases (infections of the upper and lower respiratory tract, urinary and biliary tract, soft tissue infections, etc.) - 4-6 million units/day for 4 administrations. For severe infections (sepsis, septic endocarditis, meningitis, etc.) - 10-20 million units/day; with gas gangrene - up to 40-60 million units/day.
Daily dose for children under 1 year of age - 50-100 thousand units/kg, over 1 year of age - 50 thousand units/kg; if necessary - 200-300 thousand units/kg, for “vital” indications - increase to 500 thousand units/kg. Frequency of administration: intramuscularly - 4-6 times a day, intravenously - 1-2 times a day in combination with intramuscular injections.
Subcutaneously for infiltrating infiltrates at a concentration of 100-200 thousand units in 1 ml of 0.25-0.5% procaine solution.
In the cavity (abdominal, pleural, etc.) solution of benzylpenicillin and sodium salt is administered to adults at a concentration of 10-20 thousand units per 1 ml, for children - 2-5 thousand units per 1 ml. Water for injection or 0.9% sodium chloride solution is used as a solvent. The duration of treatment is 5-7 days, followed by switching to intramuscular administration.
For eye diseaseseye drops containing 20-100 thousand units in 1 ml of sterile 0.9% sodium chloride solution or distilled water are prescribed. Administer 1-2 drops 6-8 times a day. The solution is used freshly prepared
For ear drops or drops in the nosesolutions containing 10-100 thousand units in 1 ml of sterile 0.9% sodium chloride solution or distilled water are used. Administer 1-2 drops 6-8 times a day.
The duration of treatment with benzylpenicillin, depending on the form and severity of the disease, is 7-10 days.
Method of preparing solutions
Solutions are used immediately after preparation, avoiding the addition of other medications to them.
For intramuscular injection add 1-3 ml of water for injection, 0.9% sodium chloride solution or 0.5% procaine (novocaine) solution to the contents of the bottle. The resulting solution is injected deep into the muscle.
When benzylpenicillin a is diluted in a procaine solution, turbidity of the solution may be observed due to the formation of benzylpenicillin procaine crystals, which is not an obstacle to intramuscular and subcutaneous administration drug.
For intravenous jet administration single dose(1-2 million units) are dissolved in 5-10 ml of sterile water for injection or 0.9% sodium chloride solution and administered slowly over 3-5 minutes.
For intravenous drip administration, 2-5 million units are diluted with 100-200 ml of 0.9% sodium chloride solution or 5-10% dextrose solution and administered at a rate of 60-80 drops/min. When administered dropwise to children, a 5-10% dextrose solution (30-100 ml depending on the dose and age) is used as a solvent.
For subcutaneous administration, the contents of the bottle are diluted in a 0.25-0.5% procaine solution: 500 thousand units in 2.5-5 ml, 1 million units in 5-10 ml, respectively.
For intracavitary administration, the contents of the bottle are diluted in 0.9% sodium chloride solution or in water for injection: adults - 500 thousand units in 25-50 ml, 1 million units in 50-100 ml, respectively, children - 500 thousand units in 100-250 ml, 1 million units in 200-500 ml, respectively.
Eye drops should be prepared ex tempore: the contents of the bottle are diluted in a 0.9% solution of sodium chloride or distilled water: 500 thousand units in 5-25 ml, 1 million units in 10-50 ml, respectively.
Release form
Release form
Powder for the preparation of solution for injection and local use 50,000 units, 1,000,000 units.
500,000 units and 1,000,000 units each active substance in bottles with a capacity of 10 ml or 20 ml, hermetically sealed with rubber stoppers, crimped aluminum caps or combined aluminum caps with plastic caps.
1, 5, 10 bottles along with instructions for use are placed in a cardboard pack.
50 bottles are placed in a cardboard box with an equal number of instructions for use attached for delivery to hospitals.
Storage conditions
In a dry place at a temperature of 15 to 25°C. Keep out of the reach of children.
Best before date
3 years. Do not use after the expiration date stated on the package.
Instructions for use of benzylpenicillin sodium salt for injection
for the treatment of animals with diseases of bacterial etiology
(developer organization: CJSC NPP "Agropharm", Voronezh)
I. General information
Trade name medicinal product: Benzylpenicillin sodium salt for injection (Benzylpenicillium natrium pro injectionibus).
International generic name: benzylpenicillin sodium salt.
Dosage form: powder for the preparation of solution for injection.
The drug as active substance contains benzylpenicillin sodium salt - not less than 1650 units/mg (in terms of dry matter).
By appearance the drug is a fine-crystalline powder of white or slightly yellowish color.
The drug is produced in the form of a sterile powder, packaged in 1,000,000 units in bottles sealed with rubber stoppers reinforced with aluminum caps.
Store the drug in the manufacturer's sealed packaging in a dry place, protected from light, separately from food and feed, at a temperature of 5°C to 25°C.
The shelf life of the medicinal product, subject to storage conditions, is 3 years from the date of production.
After opening the bottle, the drug cannot be stored.
Do not use after expiration date.
Benzylpenicillin sodium salt for injection should be kept out of the reach of children.
Unused medicinal product is disposed of in accordance with legal requirements.
II. Pharmacological properties
Benzylpenicillin sodium salt belongs to antibacterial drugs from the group of β-lactam antibiotics.
Benzylpenicillin is active against gram-positive microorganisms (streptococci, staphylococci, pneumococci, enterococci, most anaerobes, actinomycetes, clostridia, anthrax bacillus), some gram-negative cocci (gonococci, meningococci), as well as spirochetes. Ineffective against most gram-negative bacteria, rickettsia, viruses, protozoa, fungi, and microorganisms that produce penicillinase.
Mechanism bactericidal action is based on disruption of the synthesis of peptidoglycan, which is part of the cell wall of microorganisms, by inhibiting the enzymes transpeptidase and carboxypeptidase, which leads to disruption of the osmotic balance and destruction of the bacterial cell.
Benzylpenicillin, when administered intramuscularly, is quickly absorbed from the injection site into the blood and easily penetrates into most organs and tissues of the body. The maximum concentration of the antibiotic in the blood is achieved after 30-60 minutes, the therapeutic concentration in organs and tissues is maintained for 4-6 hours. Benzylpenicillin is excreted from the body unchanged, mainly in the urine and in small quantities with bile, in lactating animals partially with milk.
Benzylpenicillin sodium salt, according to the degree of impact on the body, according to GOST 12.1.007-76, is classified as a moderately hazardous substance (hazard class 3), and in recommended doses does not have a local irritant effect.
III. Application procedure
Benzylpenicillin sodium salt for injection is used in farm animals, fur animals and dogs for the treatment of pasteurellosis, bronchopneumonia, diseases of the genitourinary system, mastitis, necrobacteriosis, streptococcal septicemia, staphylococcosis, actinomycosis, emphysematous carbuncle, wound and postpartum sepsis, as well as other primary and secondary infections caused by pathogens sensitive to penicillins.
A contraindication to the use of the drug is the animal’s increased individual sensitivity to antibiotics. penicillin group.
Before use, the drug in the bottle is pierced with a needle and the cap and stopper of the bottle are dissolved in 5-10 ml of water for injection or sterile isotonic sodium chloride solution.
The medicinal solution is prepared immediately before administration; it cannot be stored or heated.
The drug is administered to animals intramuscularly at intervals of 4-6 hours for 5-7 days, with severe forms illness - up to 10 days, in the doses indicated in the table (based on units of active substance per 1 kg of animal weight):
*Note: young animals of large cattle and pigs - up to 6 one month old, small cattle - up to 4 months of age, horses, dogs and fur-bearing animals - up to 1 year.
In case of an overdose of the drug, the animal may experience neurotoxic symptoms (nausea, vomiting, increased reflex excitability). In these cases, the use of the drug is stopped and the animal is prescribed symptomatic therapy.
No specific effects of the drug upon its first use or discontinuation have been identified.
You should avoid skipping the next dose of the drug, as this may lead to a decrease in its therapeutic effectiveness. If one dose is missed, the use of the drug is resumed at the same dosage and according to the same regimen.
When using benzylpenicillin sodium salt for injection in accordance with these instructions side effects and complications in animals, as a rule, are not observed. With increased individual sensitivity of the animal to antibiotics penicillin series Animals sometimes exhibit rapidly developing allergic reactions (urticaria, diarrhea, angioedema, anaphylactic shock). In this case, the use of the drug is stopped and desensitizing therapy is carried out.
It is not allowed to use Benzylpenicillin sodium salt for injection simultaneously with bacteriostatic antibiotics (aminoglycosides, tetracyclines, chloramphenicol), adrenaline, euphilin, ascorbic acid, B vitamins and non-steroidal anti-inflammatory drugs, as well as mixing in the same syringe with other drugs.
Slaughter of animals for meat is permitted no earlier than 3 days after the last use of benzylpenicillin sodium salt for injection.
Meat from animals forcedly killed before the expiration of the specified period can be used as feed for carnivorous animals.
Milk obtained from animals during the treatment period and within 24 hours after the last administration of the drug must not be used for food purposes. After heat treatment, such milk can be used as animal feed.
IV. Personal prevention measures
When conducting therapeutic measures with the use of benzylpenicillin sodium salt for injection should be observed general rules personal hygiene and safety precautions provided for when working with medicines. While working with the drug, do not drink, smoke or eat. After finishing work, wash your hands with warm water and soap.
People with hypersensitivity to penicillin antibiotics should avoid direct contact with benzylpenicillin sodium salt for injection. If the drug accidentally comes into contact with the skin, it must be washed off immediately with soap and water; if it gets into the eyes, rinse them for several minutes with running water. If allergic reactions or if the drug accidentally enters the human body, you should immediately contact medical institution(bring with you the instructions for use of the drug or the label).
The use of empty drug bottles for household purposes is prohibited.
Manufacturer: CJSC NPP "Agropharm", Russia, 394087, Voronezh region, Voronezh, st. Lomonosova, 114-b.
With the approval of this instruction, the instructions for the use of benzylpenicillin sodium salt for injection, approved by Rosselkhoznadzor on June 29, 2006, become invalid.
for medical use
medicine
BENZYLPENICILLIN SODIUM SALT
Trade name
Benzylpenicillin sodium salt
International nonproprietary name
Benzylpenicillin
Dosage form
Powder for preparing a solution for intramuscular administration 1,000,000 units
Composition per bottle
active substance: benzylpenicillin sodium salt - 1,000,000 units
Description
White powder or white powder with a slightly yellowish tint, prone to clumping, forming a stable suspension when water is added.
Pharmacotherapeutic group
Antibacterial drugs for systemic use. Penicillins penicillinase sensitive
ATS code J01SE01
Pharmacological properties
Pharmacokinetics
The maximum concentration in blood plasma after intramuscular administration is achieved after 20-30 minutes. The half-life of the drug is 30-60 minutes, in case of renal failure 4-10 hours or more. Communication with plasma proteins - 60%. Penetrates into organs, tissues and biological fluids, except for cerebrospinal fluid, eye and prostate tissues. With inflammation of the meningeal membranes, it penetrates the blood-brain barrier. Passes through the placenta and penetrates into breast milk. It is excreted unchanged by the kidneys.
Pharmacodynamics
Bactericidal antibiotic from the group of biosynthetic (“natural”) penicillins. Inhibits the synthesis of the cell wall of microorganisms. Active against gram-positive pathogens: staphylococci (which do not form penicillinase), streptococci, pneumococci, corynebacteria diphtheria, anaerobic spore-forming bacilli, anthrax bacilli, Actinomyces spp.; gram-negative microorganisms: cocci (Neisseria gonorrhoeae, Neisseria meningitidis), as well as spirochetes.
Not active against most gram-negative bacteria (including Pseudomonas aeruginosa), Rickettsia spp., protozoa. Staphylococcus spp., which produce penicillinase, are resistant to the drug.
Indications for use
Croupous and focal pneumonia, pleural empyema, bronchitis
Sepsis, septic endocarditis (acute and subacute)
Peritonitis
Meningitis
Osteomyelitis
Pyelonephritis, pyelitis, cystitis, urethritis, gonorrhea, blenorrhea, syphilis, cervicitis
Cholangitis, cholecystitis
Wound infection
Erysipelas, impetigo, secondary infected dermatoses
Diphtheria
Scarlet fever
Anthrax
Actinomycosis
Sinusitis, otitis media
Purulent conjunctivitis
Directions for use and doses
Benzylpenicillin sodium salt is administered intramuscularly. When administered intramuscularly, single doses for moderate disease (infections of the upper and lower respiratory tract, urinary and biliary tract, soft tissue infections, etc.) are 250,000 - 500,000 units 4-6 times a day. For severe infections (sepsis, septic endocarditis, meningitis, etc.) - 10-20 million units per day; with gas gangrene - up to 40-60 million units.
The daily dose for children under 1 year of age is 50,000 - 100,000 units/kg, over 1 year of age - 50,000 units/kg; if necessary - 200,000 - 300,000 IU/kg, for health reasons - increase to 500,000 IU/kg. The frequency of administration is 4-6 times a day.
A solution of the drug for intramuscular administration is prepared immediately before administration by adding 1-3 ml of water for injection, 0.9% sodium chloride solution or 0.5% procaine (novocaine) solution to the contents of the bottle. When benzylpenicillin is dissolved in a procaine solution, cloudiness of the solution may be observed due to the formation of benzylpenicillin procaine crystals, which is not an obstacle to intramuscular administration of the drug. The resulting solution is injected deep into the muscle. The duration of treatment, depending on the severity of the disease, is from 7 to 10 days.
Side effects
Violation of the pumping function of the myocardium, arrhythmias, cardiac arrest, chronic heart failure (since hypernatremia may occur when administered in large doses)
Nausea, vomiting, stomatitis, glossitis, liver dysfunction
Renal dysfunction
Anemia, leukopenia, thrombocytopenia
Increased reflex excitability, meningeal symptoms, convulsions, coma
- allergic reactions: sometimes - hyperthermia, urticaria, skin rash, fever, chills, increased sweating, rash on mucous membranes, arthralgia, eosinophilia, angioedema, interstitial nephritis, bronchospasm, rarely - anaphylactic shock
- local reactions: pain and hardness at the site of intramuscular injection
Dysbacteriosis, development of superinfection (with long-term use)
Contraindications
Hypersensitivity to penicillin and other ß-lactam antibiotics
Endolumbar administration for epilepsy.
Drug interactions
Antacids, glucosamine, laxatives, aminoglycosides slow down and reduce the absorption of benzylpenicillin sodium salt. Ascorbic acid at joint use increases the absorption of benzylpenicillin sodium salt.
Bactericidal antibiotics (including cephalosporins, vancomycin, rifampicin, aminoglycosides) have a synergistic effect; bacteriostatic (including macrolides, chloramphenicol, lincosamides, tetracyclines) - antagonistic. Benzylpenicillin sodium salt increases effectiveness indirect anticoagulants(by suppressing intestinal microflora, reduces prothrombin index); reduces the effectiveness of oral contraceptives, drugs, during the metabolism of which para-aminobenzoic acid is formed.
Diuretics, allopurinol, tubular secretion blockers, phenylbutazone, non-steroidal anti-inflammatory drugs, reducing tubular secretion, increase the concentration of benzylpenicillin sodium salt.
Allopurinol increases the risk of allergic reactions (skin rash).
Special instructions
With caution: pregnancy, lactation period, allergic diseases(bronchial asthma, hay fever), renal failure.
Solutions of the drug are prepared immediately before administration. If no effect is observed after 2-3 days (maximum 5 days) after starting to use the drug, you should switch to the use of other antibiotics or combination therapy. Due to the possibility of developing fungal infections, it is advisable to long-term treatment benzylpenicillin to prescribe B vitamins, and, if necessary, antifungal drugs. It must be taken into account that the use of insufficient doses of the drug or too early termination treatment often leads to the emergence of resistant strains of pathogens.
Pregnancy and lactation
The use of the drug during pregnancy is possible only if the expected benefit to the mother outweighs the potential risk to the fetus. If it is necessary to prescribe the drug during lactation, breastfeeding should be stopped.
Features of the effect of the drug on the ability to drive vehicle or potentially dangerous mechanisms
During administration of the drug, caution should be exercised when driving vehicles, using machinery, and performing other potentially dangerous species activities that require increased concentration and speed of psychomotor reactions.
Overdose
Dosage form:powder for solution for injection and topical use
Compound:Active substance:
Benzylpenicillin sodium (benzylpenicillin sodium salt) - 500,000 units and 1,000,000 units. Description:White powder.
Pharmacotherapeutic group:Antibiotic - biosynthetic penicillin ATX:S.01.A.A.14 Benzylpenicillin
Pharmacodynamics:Bactericidal antibiotic from the group of biosynthetic (“natural”) penicillins. Suppresses the synthesis of the cell wall of microorganisms. Active against gram-positive pathogens: Staphylococcus spp. (non-penicillinase-forming), Streptococcus spp. (including Streptococcus pneumoniae), Corynebacterium spp. (including Corynebacterium diphtheriae), Bacillus anthracis, Actinomyces spp.; gram-negative microorganisms: Neisseria gonorrhoeae, Neisseria meningitidis, as well as against Treponema spp., class Spirochaetes. Not active against most gram-negative bacteria (including Pseudomonas aeruginosa), Rickettsia spp., viruses, protozoa. Staphylococcus spp., which produce penicillinase, are resistant to the drug.
Pharmacokinetics:The time required to achieve maximum concentration in blood plasma when administered intramuscularly is 20-30 minutes. Communication with plasma proteins - 60%. Penetrates into organs, tissues and biological fluids, except for cerebrospinal fluid, eye and prostate tissues; during inflammation of the meningeal membranes, it penetrates through blood-brain barrier. It is excreted unchanged by the kidneys. The half-life is 30-60 minutes, in case of renal failure - 4-10 hours or more.
Indications:Bacterial infections caused by penicillin-sensitive pathogens: community-acquired pneumonia, pleural empyema, bronchitis; sepsis, septic endocarditis (acute and subacute), peritonitis; meningitis; osteomyelitis; infections of the genitourinary system (pyelonephritis, pyelitis, cystitis, urethritis, cervicitis), biliary tract (cholangitis, cholecystitis); wound infection, skin and soft tissue infections: erysipelas, impetigo, secondary infected dermatoses; diphtheria; scarlet fever; anthrax; actinomycosis; infections of the ENT organs, eye diseases (acute conjunctivitis, corneal ulcer, etc.); gonorrhea, syphilis.
Contraindications:Hypersensitivity, including to other penicillins, cephalosporins.
With caution:Pregnancy, allergic diseases (bronchial asthma, hay fever), renal failure.
Pregnancy and lactation:The use of the drug during pregnancy is possible only if the expected benefit to the mother outweighs the potential risk to the fetus.
If it is necessary to prescribe the drug during lactation, breastfeeding should be stopped.
Directions for use and dosage:Benzylpenicillin sodium salt is administered intramuscularly, intravenously, subcutaneously, locally.
Intravenously and intramuscularly:
for moderate disease (infections of the upper and lower respiratory tract, urinary and biliary tract, infection of soft tissues, etc.) - 4-6 million units/day for 4 administrations. For severe infections (sepsis, septic endocarditis, meningitis, etc.) - 10-20 million units/day; with gas gangrene - up to 40-60 million units/day.The daily dose for children under 1 year of age is 50-100 thousand units/kg, over 1 year of age - 50 thousand units/kg; if necessary - 200-300 thousand units/kg, for "vital" indications - increase to 500 thousand units/kg. Frequency of administration: intramuscularly - 4-6 times a day, intravenously - 1-2 times a day in combination with intramuscular injections.
Subcutaneously for infiltrating infiltrates at a concentration of 100-200 thousand units in 1 ml of 0.25-0.5% procaine solution.
In the cavity (abdominal, pleural, etc.) a solution of benzylpenicillin sodium salt is administered to adults at a concentration of 10-20 thousand units per 1 ml, for children - 2-5 thousand units per 1 ml. The solvent used for injection is 0.9% sodium chloride solution. The duration of treatment is 5-7 days, followed by switching to intramuscular administration.
For eye diseases appoint eye drops , containing 20-100 thousand units in 1 ml of sterile 0.9% sodium chloride solution or distilled water. Inject 1-2 drops 6-8 times a day. The solution is used freshly prepared.
For ear drops or drops in the nose solutions containing 10-100 thousand units in 1 ml of sterile 0.9% sodium chloride solution or distilled water are used. Administer 1-2 drops 6-8 times a day.
The duration of treatment with benzylpenicillin, depending on the form and severity of the disease, is 7-10 days.
Solutions are used immediately after preparation, avoiding the addition of other medications to them.
For intramuscular administration add 1-3 ml of water for injection, 0.9% sodium chloride solution or 0.5% procaine (novocaine) solution to the contents of the bottle. The resulting solution is injected deep into the muscle.
When diluting benzylpenicillin in a solution of procaine, cloudiness of the solution may be observed due to the formation of procaine crystals, which is not an obstacle to intramuscular and subcutaneous administration of the drug.
For intravenous jet administration a single dose (1-2 million units) is dissolved in 5-10 ml of sterile water for injection or 0.9% sodium chloride solution and administered slowly over 3-5 minutes.
For intravenous drip administration 2-5 million units are diluted in 100-200 ml of 0.9% sodium chloride solution or 5-10% dextrose solution and administered at a rate of 60-80 drops/min. When administered dropwise to children, a 5-10% dextrose solution (30-100 ml depending on the dose and age) is used as a solvent.
For subcutaneous administration the contents of the bottle are diluted in 0.25-0.5% procaine solution: 500 thousand units in 2.5-5 ml, 1 million units in 5-10 ml, respectively.
For intracavitary administration the contents of the bottle are diluted in 0.9% sodium chloride solution or water for injection: adults - 500 thousand units in 25-50 ml, 1 million units in 50-100 ml, respectively; for children - 500 thousand units in 100-250 ml, 1 million units in 200-500 ml, respectively.
Eye drops should be prepared ex tempore: the contents of the bottle are diluted in a 0.9% solution of sodium chloride or distilled water: 500 thousand units in 5-25 ml, 1 million units in 10-50 ml, respectively.
Ear drops and nasal drops: the contents of the bottle are diluted in 0.9% sodium chloride solution or distilled water: 500 thousand units in 5-50 ml, 1 million units in 10-100 ml, respectively.
Side effects:Allergic reactions: hyperthermia, urticaria, skin rash, rash on mucous membranes, arthralgia, eosinophilia, angioedema, interstitial nephritis, bronchospasm, anaphylactic shock. At the beginning of the course of treatment (especially when treating congenital syphilis) - fever, chills, increased sweating, exacerbation of the disease, Jarisch-Herxheimer reaction.
From the outside cardiovascular system:
violation of the pumping function of the myocardium, arrhythmias, cardiac arrest, chronic heart failure (since hypernatremia may occur when large doses are administered).Local reactions: soreness and hardness at the site of intramuscular injection.
With long-term use: dysbacteriosis, development of superinfection.
If any of the side effects indicated in the instructions get worse, or you notice any other side effects not listed in the instructions, please inform your doctor.
Overdose:Manifests toxic effect to the central nervous system(convulsions, headache, myalgia, arthralgia).
Treatment is symptomatic.
Interaction:Bactericidal antibiotics (including cephalosporins, aminoglycosides) have a synergistic effect; bacteriostatic (including macrolides, lincosamides, tetracyclines) - antagonistic. Increases the effectiveness of indirect anticoagulants (suppressing intestinal microflora, reduces the prothrombin index); reduces the effectiveness of oral contraceptives, drugs, during the metabolism of which para-aminobenzoic acid is formed.
Diuretics, tubular secretion blockers, non-steroidal anti-inflammatory drugs, reducing tubular secretion, increase the concentration of benzylpenicillin.
Allopurinol increases the risk of allergic reactions (skin rash).
Special instructions:Solutions of the drug are prepared immediately before administration. If no effect is observed after 2-3 days (maximum 5 days) after starting to use the drug, you should move on to the use of other antibiotics or combination therapy. Due to the possibility of developing fungal infections, it is advisable to prescribe B vitamins and, if necessary, antifungal drugs during long-term treatment with benzylpenicillin. It must be taken into account that the use of insufficient doses of the drug or stopping treatment too early often leads to the emergence of resistant strains of pathogens. Impact on the ability to drive vehicles. Wed and fur.:During the administration of the drug, care should be taken when driving vehicles, machinery and when performing other potentially dangerous types of activities that require increased concentration and speed of psychomotor reactions.
1 bottle of powder for preparation injection solution contains 0.6 g (1 million IU) or 3 g (5 million IU) benzylpenicillin in the form of sodium salt, in packs of 1 and 50 pcs.
Pharmacological action
Pharmacological action- bactericidal, antibacterial.Indications for the drug Benzylpenicillin
Severe generalized infections (meningitis, septicemia, disseminated gonococcal infection), streptococcal endocarditis, congenital syphilis, abscesses, respiratory tract infections (pneumonia), skin infections (erysipelas), lymphadenitis and lymphangitis, osteomyelitis, anthrax, tetanus, gas gangrene.
Contraindications
History of hypersensitivity to penicillins.
Side effects
Allergic reactions (acute hypersensitivity reactions occur 1-30 minutes after taking the drug), damage to the central nervous system (convulsions) with rapid injection of high doses.
Interaction
Probenecid, by reducing renal excretion, increases blood levels.
Directions for use and doses
Dosage varies depending on the type and severity of the infection; Approximate recommended doses: adults - 1.8-3.6 g (3-6 million IU) per day in 4 (6) divided doses. For severe infections (meningitis, septicemia) - up to 200 (300) mg/kg (0.33-0.5 million IU/kg) per day in 4-6 divided doses. In these cases, treatment begins with a slow intravenous injection or a rapid infusion (20-40 ppm, using 5% glucose as a diluent). Children: 30-90 mg/kg (50,000-150,000 IU/kg) per day in 4-6 divided doses. This corresponds to approximately 240 mg 4 times daily for children 5 months to 2 years; 300-600 mg 4 times a day for children from 2 to 6 years old and 450-900 mg 4 times a day for children 7-12 years old. For severe infections (meningitis, septicemia) - up to 300 mg/kg (0.5 million IU/kg) per day in 4-6 doses. In these cases, treatment begins with a slow intravenous injection or a rapid infusion (20-40 minutes, using 5% glucose as a diluent). Benzylpenicillin can also be used intramuscularly. Dilution of benzylpenicillin for infusion should be carried out immediately before use, since the activity of the antibiotic quickly decreases in infusion solutions. The duration of treatment depends on the indications and clinical picture. Recommended doses for individual diseases are: meningitis and/or septicemia caused by meningococci - 180-240 mg/kg (0.3-0.4 million IU/kg) per day in 4-6 doses, in the form of a slow IV injection or infusion over minimum 5 days; meningitis and/or septicemia caused by pneumococci - 240-300 mg/kg (0.4-0.5 million IU/kg) per day in 4-6 divided doses, as a slow IV injection or infusion over at least 10 days; infections caused by clostridia - 9-12 g (15-20 million IU) per day for adults and 180 mg/kg (0.3 million IU/kg) per day for 1 week as an addition to antitoxin therapy; streptococcal endocarditis - 6-12 g (10-20 million IU) per day for adults and 180 mg/kg (0.3 million IU) - for children for 2-4 weeks; congenital syphilis - 30 mg/kg (50,000 IU/kg) per day IM or IV in 2 doses for 2 weeks.
Storage conditions for the drug Benzylpenicillin
In a place protected from light, at a temperature not exceeding 30 °C.Keep out of the reach of children.
Shelf life of the drug Benzylpenicillin
4 years. 4 years; prepared solution for injection 24 hours (in the refrigerator 72 hours), for infusion - 12 hours (in the refrigerator - 24 hours).Do not use after the expiration date stated on the packaging.
Instructions for medical use
Synonyms of nosological groups
| ICD-10 rubric | Synonyms of diseases according to ICD-10 |
|---|---|
| A35 Other forms of tetanus | Hydrophobia (hydrophobia) |
| Clostridia wounds | |
| Muscle spasms due to tetanus | |
| Tetanus | |
| Tetanus local | |
| Tetanus | |
| A41.9 Septicemia, unspecified | Bacterial septicemia |
| Severe bacterial infections | |
| Generalized infections | |
| Generalized systemic infections | |
| Generalized infections | |
| Wound sepsis | |
| Septic-toxic complications | |
| Septicopyemia | |
| Septicemia | |
| Septicemia/bacteremia | |
| Septic diseases | |
| Septic conditions | |
| Septic shock | |
| Septic condition | |
| Toxic-infectious shock | |
| Septic shock | |
| Endotoxin shock | |
| A46 Erysipelas | Erysipelas |
| A53.9 Syphilis, unspecified | Syphilis |
| Tertiary syphilis | |
| A54 Gonococcal infection | Gonococcal infections |
| Disseminated gonococcal infection | |
| Disseminated gonorrheal infection | |
| B99 Other infectious diseases | Infections (opportunistic) |
| Infections due to immunodeficiency | |
| Opportunistic infections | |
| I33 Acute and subacute endocarditis | Postoperative endocarditis |
| Early endocarditis | |
| Endocarditis | |
| Endocarditis acute and subacute | |
| I88 Nonspecific lymphadenitis | Lymphadenitis |
| Lymphadenitis of nonspecific etiology | |
| Superficial lymphadenitis | |
| J06 Acute infections upper respiratory tract of multiple and unspecified localization | Bacterial infections of the upper respiratory tract |
| Bacterial respiratory infections | |
| Pain due to colds | |
| Pain in infectious and inflammatory diseases of the upper respiratory tract | |
| Viral respiratory disease | |
| Viral respiratory tract infections | |
| Inflammatory disease of the upper respiratory tract | |
| Inflammatory diseases of the upper respiratory tract | |
| Inflammatory diseases of the upper respiratory tract with difficult to separate sputum | |
| Inflammatory diseases of the respiratory tract | |
| Secondary infections with influenza | |
| Secondary infections due to colds | |
| Influenza conditions | |
| Difficulty secreting sputum in acute and chronic diseases respiratory tract | |
| Upper respiratory tract infections | |
| Upper respiratory tract infections | |
| Respiratory tract infections | |
| Respiratory and lung infections | |
| ENT infections | |
| Infectious and inflammatory diseases of the upper respiratory tract | |
| Infectious and inflammatory diseases of the upper respiratory tract and ENT organs | |
| Infectious and inflammatory diseases of the upper respiratory tract in adults and children | |
| Infectious and inflammatory diseases of the upper respiratory tract | |
| Infectious inflammation of the respiratory tract | |
| Respiratory tract infection | |
| Qatar of the upper respiratory tract | |
| Catarrhal inflammation of the upper respiratory tract | |
| Catarrhal disease of the upper respiratory tract | |
| Catarrhal phenomena from the upper respiratory tract | |
| Cough in diseases of the upper respiratory tract | |
| Cough with a cold | |
| Fever due to influenza | |
| ARVI | |
| acute respiratory infections | |
| Acute respiratory infection with symptoms of rhinitis | |
| Acute respiratory infection | |
| Acute infectious-inflammatory disease of the upper respiratory tract | |
| Acute cold | |
| Acute respiratory disease | |
| Acute respiratory disease of influenza nature | |
| Sore throat or nose | |
| Cold | |
| Colds | |
| Colds | |
| Respiratory infection | |
| Respiratory viral infections | |
| Respiratory diseases | |
| Respiratory infections | |
| Recurrent respiratory tract infections | |
| Seasonal colds | |
| Seasonal colds | |
| Frequent colds and viral diseases | |
| J85 Abscess of the lung and mediastinum | Lung abscess |
| Lung abscess | |
| Bacterial lung destruction | |
| L02 Skin abscess, boil and carbuncle | Abscess |
| Skin abscess | |
| Carbuncle | |
| Skin carbuncle | |
| Furuncle | |
| Skin boil | |
| Furuncle of the external auditory canal | |
| Furuncle of the auricle | |
| Furunculosis | |
| Boils | |
| Chronic recurrent furunculosis | |
| M60.0 Infectious myositis | Muscle abscess |
| Soft tissue infections | |
| Infectious myositis | |
| Pyomyositis | |
| Specific infectious processes in soft tissues | |
| M65.0 Tendon sheath abscess | Soft tissue infections |
| M65.1 Other infectious tenosynovitis | Soft tissue infections |
| Tenosynovitis infectious | |
| M71.0 Abscess of bursa | Soft tissue infections |
| M71.1 Other infectious bursitis | Bacterial bursitis |
| Infectious bursitis | |
| Soft tissue infections | |
| N74.2 Inflammatory diseases of women pelvic organs caused by syphilis (A51.4+, A52.7+) | Syphilis |
| N74.3 Gonococcal inflammatory diseases female pelvic organs (A54.2+) | Gonorrheal diseases |
| Gonorrhea | |
| Gonococcal urethritis | |
| R09.1 Pleurisy | Calcification of the pleura |
| Acute pleurisy | |
| R78.8.0* Bacteremia | Bacteremia |
| Persistent bacteremia |