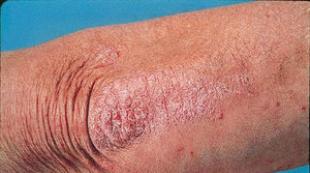
Bilateral total pneumonia acinetobacter baumannii. Infections caused by ACINETOBACTER BAUMANNII: risk factors, diagnosis, treatment, approaches to prevention
Infections caused by Acinetobacter baumannii: risk factors, diagnosis, treatment, approaches to prevention /
Belarusian State Medical University Research Institute of Antimicrobial Chemotherapy, Smolensk State Medical Academy, Russian Federation
Gorbich U.L., Karpov I.A., Krechikova O.I.
Infections induced byAcinetobacter baumannii: risk factors, diagnostics, treatment, prevention approaches
Nosocomial infections (lat. nosocomium- hospital, Greek nosocmeo- hospital, care for the patient) are infections that developed in the patient at least 48 hours after hospitalization, provided that upon admission to the hospital the infection did not exist and was not in the incubation period; infections resulting from previous hospitalization, as well as infectious diseases of medical workers associated with their professional activities.
According to various authors, the number of patients who develop nosocomial infections ranges from 3 to 15% ?. Of these, 90% are of bacterial origin; viral, fungal pathogens and protozoa are much less common.
From the beginning of the era of antibiotics until the 60s of the twentieth century. approximately 65% of nosocomial infections (HAIs) were staphylococcal in nature. With the advent of penicillinase-stable antibacterial drugs in the arsenal of doctors, they receded into the background, giving way to infections caused by gram-negative bacteria.
Currently, despite the slightly increased etiological role of gram-positive microorganisms and fungi as causative agents of nosocomial infections, strains of gram-negative microorganisms with multiple resistance to antibacterial drugs pose a serious problem in hospitals around the world. According to a number of authors, their frequency varies from 62 to 72% of all nosocomial infections. The most relevant pathogens of all nosocomial infections (except angiogenic ones) and sepsis are microorganisms of the family Enterobacteriaceae and non-fermenting bacteria, which include Pseudomonasaeruginosa And Acinetobacterspp. .
The most clinically significant species of the genus Acinetobacter is Acinetobacter baumannii(genomospecies 2), which causes 2-10% of gram-negative infections in Europe and the United States, up to 1% of all nosocomial infections.
Risk factors
As common risk factors for infections caused by A. baumannii, highlight:
Male gender;
Old age;
The presence of concomitant diseases (malignant blood diseases, cardiovascular or respiratory failure, disseminated intravascular coagulation);
Duration of use of invasive methods of treatment and monitoring (ventilation for more than 3 days; inhalation administration of drugs; insertion of a nasogastric tube; tracheostomy; catheterization of the bladder, central vein, artery, surgery);
Long-term stay in a hospital or intensive care unit (ICU);
Previous antibacterial therapy using cephalosporins, fluoroquinolones or carbapenems.
Surgery prior to ICU admission increases the risk of infection by approximately 5 times.
As risk factors for infection with a carbapenem-resistant strain A. baumannii for adults, the following have been described so far: large hospital size (more than 500 beds); hospitalization in the ICU or hospitalization for emergency reasons; long hospital stay; high density of CRAB patients in the ward; male gender; immunosuppression; Mechanical ventilation, catheterization of the urinary tract or artery, hemodialysis; recent surgery; pulse lavage of wounds; previous use of meropenem, imipenem or ceftazidime.
In the Republic of Belarus, as risk factors for colonization/infection with a nosocomial isolate Acinetobacter baumannii resistant to carbapenem antibiotics were identified by previous use of “antipseudomonas” carbapenems, urinary tract catheterization, hospitalization in a non-therapeutic department and age under 40 years (Table 1).
Table 1 Risk factors for colonization/infection with a carbapenem-resistant strain A. baumannii in hospital healthcare organizations in Minsk(personal unpublished data)
* Odds ratio (OR) - defined as the ratio of the odds of an event in one to the odds of an event in the other, or as the ratio of the odds that an event will occur to the odds that an event will not occur; ** meropenem, imipenem, doripenem.
Acinetobacter-associated
infections
A. baumannii in most cases it causes disease in seriously ill immunocompromised patients. This microorganism can cause infections of the respiratory tract (sinusitis, tracheobronchitis, pneumonia), bloodstream (sepsis, endocarditis of natural and artificial valves), urinary tract, wound and surgical infections, skin and soft tissue infections (including necrotizing fasciitis), nervous system (meningitis , ventriculitis, brain abscess), intra-abdominal (abscesses of various locations, peritonitis), musculoskeletal system (osteomyelitis, arthritis).
According to our own research conducted in 15 hospital healthcare organizations in Minsk, in the structure A. baumannii-associated infections, bloodstream infections predominate, accounting for 39.4% of all infections caused by this pathogen. The second place is occupied by respiratory tract infections (35.4%), the third (19.7%) is infections of the skin and soft tissues (including infections of a surgical wound). Osteomyelitis was observed in 4.7% of cases, urinary tract infections - 0.8% of cases.
Bloodstream infections. Clinical manifestations of bloodstream infections caused by A. baumannii, range from transient bacteremia to extremely severe disease with a high mortality rate. The portal of infection is most often the respiratory tract, however, during the primary development of the septic process, the main role is played by intravascular catheters. Less common portals of entry include the urinary tract, skin and soft tissue, burn wounds, abdominal organs, and central nervous system. Nosocomial sepsis caused by A. baumannii, in 73% of cases develops after the 15th day of hospitalization. Septic shock develops in approximately 30% of patients with Acinetobacter-associated sepsis. However, patients with bacteremia associated with intravascular catheters have a better prognosis, presumably because the source of infection can be eliminated from the body when the catheter is removed.
Risk factors for developing bloodstream infections caused by A. baumannii, are emergency hospitalization, long hospital stay, previous colonization with Acinetobacter, high rate of invasive procedures, mechanical ventilation, advanced age or age less than 7 days, weight less than 1500 g (for newborns), immunosuppression, malignant diseases, cardiovascular failure, renal insufficiency, respiratory failure during admission to the ICU, a history of an episode of sepsis that developed in the ICU, previous antibiotic therapy (especially ceftazidime or imipenem).
Respiratory tract infections. A. baumannii, along with Pseudomonas aeruginosa, Stenotrophomonasmaltophilia and MRSA, is the causative agent of late (developing later than 5 days from the moment of hospitalization) episodes of nosocomial pneumonia. In addition to the time of manifestation of the infection, previous antibiotic therapy and hospitalization within the last 60 days are also important.
Nosocomial Acinetobacter-associated pneumonia is most often polysegmental. The formation of cavities in the lungs, pleural effusion, and the formation of a bronchopleural fistula may be observed.
Independent risk factors for the development of VAP caused by A. baumannii, are previous antibacterial therapy and the presence of acute respiratory distress syndrome. A previous episode of sepsis, use of antibacterial drugs before the development of infection (especially imipenem, fluoroquinolones and third generation cephalosporins, piperacillin/tazobactam), duration of mechanical ventilation for more than 7 days, reintubation, length of hospital stay have been identified as risk factors for the development of VAP caused by a multidrug-resistant strain A. baumannii .
A. baumannii is the third most common cause of nosocomial tracheobronchitis (NTB) in patients on mechanical ventilation, causing 13.6 and 26.5% of cases of NTB in patients with surgical and therapeutic pathology, respectively. The development of NTB significantly led to an increase in the length of stay in the ICU and the duration of mechanical ventilation, even in cases where the patients did not subsequently develop nosocomial pneumonia.
Skin and soft tissue infections. A.baumannii is a significant pathogen in traumatic injuries, burns, as well as in relation to infectious complications of postoperative wounds. Skin and soft tissue infections caused by A. baumannii, in most cases are complicated by bacteremia.
Acinetobacters can cause infections of the subcutaneous fat at the site of an intravenous catheter, the resolution of which can only be achieved after its removal.
Nervous system infections. Acinetobacter baumannii capable of causing nosocomial meningitis and brain abscesses. Meningitis can develop acutely or have a gradual onset. A petechial rash may be observed on the skin (up to 30% of cases). Changes in cerebrospinal fluid in meningitis caused by A. baumannii, do not differ from the corresponding changes in meningitis of other etiologies and are represented by: pleocytosis with a predominance of neutrophils, an increase in protein and lactic acid levels, and a decrease in glucose levels.
Risk factors for the development of acinetobacter meningitis include: emergency neurosurgical intervention, external ventriculostomy (especially performed within 5 days), the presence of a cerebrospinal fistula, irrational use of antibacterial drugs in neurosurgical ICUs.
Urinary tract infections (UTIs). Despite frequent colonization of the lower urinary tract, Acinetobacter is rarely the etiological agent of UTI. Acinetobacter spp.. are isolated in 1-4.6% of cases of nosocomial UTI.
Risk factors for Acinetobacter -associated UTIs include the presence of a bladder catheter and nephrolithiasis.
Other infections. Acinetobacter causes peritonitis in patients receiving long-term ambulatory peritoneal dialysis; as well as cholangitis against the background of transhepatic cholangiography or biliary drainage. Osteomyelitis and arthritis caused by A. baumannii, are associated with the introduction of artificial implants or trauma. Acinetobacter-associated eye lesions associated with contamination of soft contact lenses (ulceration and perforation of the cornea) have also been described. It is possible to develop other lesions of the organ of vision from conjunctivitis to endophthalmitis.
Diagnosis and definition
sensitivity to antimicrobial drugs
In clinical practice, infections caused by A. baumannii, preceded by colonization of the skin, respiratory and urinary tract, and gastrointestinal tract of patients. Significant spread A. baumannii as a colonizing microorganism requires an objective assessment of the situation when isolated from a patient’s biological material. At the same time, it should be noted that the selection Acinetobacterspp. as a colonizing microorganism is prognostically significant for determining the etiology of subsequent nosocomial infection (positive/negative predictive value - 94/73% for VAP, 43/100% for bloodstream infections, respectively).
Diagnosis of nosocomial infection, incl. A. baumannii-associated, from a clinical point of view is conditionally divided into 4 stages:
1. Collection and transportation of clinical material.
2. Identification of the pathogen.
3. Determination of the etiological significance of the isolated microorganism.
4. Determination of sensitivity to antimicrobial drugs and interpretation of the results obtained.
Proper collection and transportation of clinical material can minimize the likelihood of unreliable laboratory results and, consequently, reduce the “inadequate” prescription of antimicrobial drugs.
General rules for collecting clinical material for microbiological testing (as amended):
1. Collection, if possible, should be carried out before the start of antibacterial therapy. If the patient is already receiving antibacterial therapy, then clinic The material should be taken immediately before the next administration of the drug.
2. Material for bacteriological research must be taken directly from the source of infection. If impossible, use other clinically significant biological material.
3. Strictly follow the rules of asepsis, avoiding contamination of the material with foreign microflora.
4. To take discharge from a wound, smears from the mucous membranes, from the eye, ear, nose, pharynx, cervical canal, vagina, anus, sterile cotton swabs should be used. For blood, pus, cerebrospinal fluid and exudates - sterile syringes and specialized transport media; for sputum, urine, feces - sterile tightly closed containers.
5. The amount of material must be sufficient to conduct the study.
6. Native material is delivered to the laboratory as soon as possible (no later than 1.5-2 hours after receipt). It is allowed to store the material in the refrigerator at 4 o C (except for biological material obtained from normally sterile loci: cerebrospinal fluid, blood, intra-articular and pleural fluid). When using transport media, clinical material can be stored for 24-48 hours.
7. Liquid biological material can be transported directly in a syringe, the tip of which is fitted with a sterile cap or an angled needle.
Pathogen identification. Genus Acinetobacter(family Moraxellaceae) consists of strict aerobic, nonmotile gram-negative lactose-non-fermenting oxidase-negative, catalase-positive coccobacteria measuring 1-1.5 x 1.5-2.5 μm, oxidizing glucose to acid only in the presence of oxygen and capable of growing on ordinary nutrient media. On solid nutrient media, colonies are smooth, opaque, and somewhat smaller in size than representatives of enterobacteria.
These microorganisms have typical morphological forms in smears made from clinical material or from liquid nutrient media. When growing on solid media in the presence of antibiotics, the bacteria in smears are rod-shaped. Some Acinetobacter isolates can retain crystal violet, bleaching poorly on Gram stains, leading to their misinterpretation as Gram-positive bacteria.
Interpretation of results(with changes and additions). It is the deep conviction of the authors that a reliable criterion for infection associated with opportunistic nosocomial microflora, including Acinetobacter baumannii, is the isolation of a culture from a sterile source.
Blood. The material for the study must be taken from at least two peripheral veins into different bottles. Blood should not be drawn from a venous catheter unless a catheter-related infection is suspected. When comparing cultures of two blood samples taken from a catheter and a peripheral vein and inoculated using a quantitative method, obtaining a colony growth from the catheter that is 5-10 times higher than the number of identical colonies from venous blood culture indicates the presence of a catheter-related infection.
Liquor. Selection A. baumannii in low concentrations makes it difficult to interpret the results, especially in departments where this microorganism often colonizes the skin of patients. The likelihood of its etiological significance increases significantly in the case of isolation of Acinetobacter from the cerebrospinal fluid of patients with an existing infection caused by A.baumannii, outside the central nervous system (so-called secondary meningitis), after neurosurgical interventions, in patients with penetrating injuries of the skull, especially against the background of existing risk factors for Acinetobacter-associated infections.
Interpretation of the clinical significance of acinetobacters isolated from non-sterile loci is a multifactorial process that depends on the qualifications of the clinician, microbiologist, specialist who collected the material, and the patient’s condition. The following criteria are to a certain extent conditional, but at the same time they make it possible to increase the likelihood of adequate interpretation of the isolated microorganism as a colonizing agent or infectious agent.
Sputum. Isolation of acinetobacters in an amount of ³ 10 6 CFU/ml (from bronchial washings ³ 10 4 CFU/ml) is diagnostically significant, provided that the rules for sputum collection are followed. However, these values are not absolute, since against the background of antibacterial therapy, the number of causally significant bacteria in the sputum decreases and, conversely, the concentration of colonizing microflora increases.
When examining sputum, bacterioscopy is mandatory, as it allows one to judge the quality of the material taken. The presence of more than 10 epithelial cells and/or less than 25 polymorphonuclear leukocytes in one field of view at low magnification indicates contamination of the sample with saliva, so further examination of this material is inappropriate. In this case, sputum should be collected again in compliance with all collection rules.
Material for wound infection. Possible contamination of the test material with isolates should be excluded A. baumannii from the surface of the skin, especially when using tampons. When isolating mixed cultures, preference should be given to microorganisms isolated in higher concentrations.
Urine. Isolation of bacteria in a concentration of ³ 10 5 CFU/ml in the presence of symptoms of the disease is diagnostically significant. When collecting urine from the bladder directly without catheterization of the urinary tract, the isolation of Acinetobacter in any titer is considered significant. The presence of three or more types of microorganisms in high concentrations indicates contamination during urine collection or improper storage.
Additional marker of etiological significance Acinetobacter baumannii is the positive dynamics of the patient’s general condition against the background of anti-Acinetobacter therapy.
Interpretation of an antibiogram(with changes and additions). After receiving the results of testing the pathogen for sensitivity to antibacterial drugs, etiotropic therapy should not be prescribed formally, relying only on the readings of the antibiogram. The body's sensitivity to a particular antimicrobial drug in vitro does not always correlate with its activity in vivo. This may be due to the individual characteristics of the pharmacokinetics and/or pharmacodynamics of the drug in this particular patient, as well as to errors in the research methodology, the quality of the materials used, etc.
When analyzing an antibiogram, you should pay attention not to any specific drug(s) to which the pathogen is sensitive/resistant, but to the whole picture. This allows, by comparing the probable resistance phenotype of acinetobacters with actual data, to correct the latter, thereby avoiding the prescription of ineffective drugs.
In particular, to identify strains that produce extended-spectrum beta-lactamases (ESBLs), attention should be paid to the sensitivity of the pathogen to cefoxitin and aztreonam. If the isolate produces an ESBL, cefoxitin remains active, but aztreonam does not. In this case, the isolate must be regarded as resistant to all I-IV generation cephalosporins and aztreonam, regardless of the actual results of the antibiogram. If a strain is resistant to cefoxitin but sensitive to aztreonam, it produces chromosomal beta-lactamases. In this case, IV generation cephalosporins can retain their activity.
If sensitivity to only one of the “antipseudomonas” carbapenems is determined, the sensitivity of the others should not be assessed by analogy with it. Different representatives of carbapenems are susceptible to varying degrees of exposure to one or another resistance mechanism. A. baumannii resistant, for example, to meropenem, may remain sensitive to imipenem and/or doripenem and vice versa.
If a strain resistant to colistin is detected, this result should be treated with caution and susceptibility should be retested with parallel testing of control strains.
With regard to aminoglycosides, interpretative assessment of the antibiotic profile is extremely difficult due to the large number of aminoglycoside-modifying enzymes and the variability of their substrate profile. Therefore, for aminoglycosides, a wide variety of sensitivity/resistance combinations within a class are acceptable.
Most clinical isolates A. baumannii is resistant to fluoroquinolones and chloramphenicol, therefore it is necessary to be careful when choosing these drugs as etiotropic drugs for the treatment of Acinetobacter-associated infections, despite the results of determining sensitivity to antibiotics. In addition, when assessing the sensitivity Acinetobacter baumannii to quinolones, one should take into account the fact that to develop resistance to non-fluorinated quinolones, one mutation in the gene of either DNA gyrase (gyrA) or topoisomerase IV (parC) is sufficient. Mutations in both genes are required for the development of resistance to fluoroquinolones. Therefore, when receiving the results of an antibiogram indicating the sensitivity of a strain to nalidixic or pipemidic acid with simultaneous resistance to fluorinated quinolones, one should be extremely skeptical about this antibiogram as a whole.
When interpreting antibiotic grams, it is also necessary to take into account that Acinetobacterspp. in general, they are naturally resistant to 1st and 2nd generation cephalosporins, natural and aminopenicillins, trimethoprim, and fosfamycin.
To characterize resistance Acinetobacter baumannii It is recommended to use the following concepts:
Resistant ( resistant) Acinetobacterbaumannii- insensitive to one antimicrobial drug;
Multidrug resistant ( multidrug- resistant - MDR) Acinetobacterbaumannii- insensitive to ³ 1 drug in ³ 3 classes listed in table. 2;
Table 2. Antimicrobials used for classification Acinetobacter spp. by degree of resistance
|
Class |
Antimicrobial drug |
|
Aminoglycosides |
Gentamicin |
|
Tobramycin |
|
|
Amikacin |
|
|
Netilmicin |
|
|
"Antipseudomonas" carbapenems |
Imipenem |
|
Meropenem |
|
|
Doripenem |
|
|
"Antipseudomonas" fluoroquinolones |
Ciprofloxacin |
|
Levofloxacin |
|
|
“Antipseudomonas” penicillins + β-lactamase inhibitors |
Piperacillin/ta-zobactam |
|
Ticarcillin/clava-lanate |
|
|
Cephalosporins |
Cefotaxime |
|
Ceftriaxone |
|
|
Ceftazidime |
|
|
Inhibitors of folic acid metabolism |
Co-trimoxazole |
|
Monobactams |
Aztreons |
|
beta-lactams + sulbactam |
Ampicillin-sul- |
|
Cefoperazone-sul- |
|
|
Polymyxins |
Colistin |
|
Polymyxin B |
|
|
Tetracyclines |
Tetracycline |
|
Doxycycline |
|
|
Minocycline |
Extensively resistant ( extensivelydrug- resistant - XDR) Acinetobacterbaumannii- insensitive to ³ 1 drug in ³ 8 classes listed in table. 2;
Pan-resistant ( pandrug- resistant - PDR) Acinetobacterbaumannii- insensitive to all listed in the table. 2 antimicrobial drugs.
When analyzing an antibiogram, no less important than the interpretation of qualitative characteristics of resistance is the assessment of the minimum inhibitory concentration (MIC). In some cases, especially if the microorganism is intermediate-resistant (i.e., the MIC value exceeds the sensitivity threshold, but does not reach the resistance threshold), based on the pharmacokinetic characteristics of the drug, it is possible to achieve a drug concentration exceeding the MIC in the site of infection when prescribing the maximum dose and/or use of a prolonged administration regimen. In particular, according to randomized controlled trials, the constant concentration of the drug achieved in serum with continuous administration is 5.8 times higher than the minimum concentration achieved with intermittent administration. And in a study by D. Wang, comparing the use of meropenem at a dose of 1 g every 8 hours intravenously for a one-hour infusion and at a dose of 0.5 g every 6 hours for a three-hour infusion in the treatment of ventilator-associated pneumonia caused by multidrug-resistant strains A. baumannii, it was found that serum drug concentrations exceeded the MIC for 54 and 75.3% of the time between doses, respectively; the cost of antibacterial therapy was significantly 1.5 times lower in the second group. In table Table 3 shows the criteria for interpreting sensitivity according to MIC and the corresponding zones of inhibition of the growth of microorganisms on a solid nutrient medium in accordance with the recommendations of the European Commission for determining sensitivity to antimicrobial drugs ( The European Committee on Antimicrobial Susceptibility Testing - EUCAST).
Table 3 Sensitivity Interpretation Criteria Acinetobacter spp.. to antimicrobial drugs according to MIC and growth retardation zones (EUCAST)
|
Antimicrobialpreparation |
MIC (mg/l) |
in disk (mcg) |
Stunted zone(mm) |
|||
|
Carbapenems |
||||||
|
Doripenem |
||||||
|
Imipenem |
||||||
|
Meropenem |
||||||
|
Fluoroquinolones |
||||||
|
Ciprofloxacin |
||||||
|
Levofloxacin |
||||||
|
Aminoglycosides |
||||||
|
Amikacin |
||||||
|
Gentamicin |
||||||
|
Netilmicin |
||||||
|
Tobramycin |
||||||
|
Colistin* |
||||||
|
Trimethoprim-sulfamethoxazole |
||||||
* Diffuses poorly into solid nutrient media. Exclusively the definition of the IPC!
Treatment
Treatment of nosocomial infections caused by Acinetobacter baumannii, is carried out in accordance with the general rules for the management of infections associated with the provision of medical care (Fig. 1). Empirical prescription of anti-Acinetobacter therapy for suspected development of nosocomial infection is justified in those healthcare organizations or their structural divisions where A. baumannii is one of the leading causative agents of these infections, taking into account risk factors.
Evaluation of the effectiveness of the therapy should be carried out 48-72 hours after its start, regardless of whether the therapy was prescribed empirically or after isolation of the pathogen. It should be based on the dynamics of the clinical picture and the results of microbiological studies (including repeated ones), and the clinical picture should serve as the prevailing factor for assessment.
Despite a number of studies indicating the possibility of reducing the duration of antibiotic therapy, the duration of antimicrobial therapy should not be reduced for infections caused by A. baumannii. Thus, in a multicenter randomized study, it was found that reducing the duration of antibacterial therapy for VAP caused by non-fermenting gram-negative microorganisms from 15 to 8 days is associated with an increase in the frequency of relapses.
When choosing therapy, it should be taken into account that throughout the world the most active antibacterial drugs against A. baumannii are sulbactam, carbapenems, aminoglycosides, polymyxins, tigecycline and minocycline. However, the choice of a specific antimicrobial drug that can be used for empirical therapy A. baumannii-associated infections should be based on local data from the department or healthcare organization where the nosocomial infection developed.
If antimicrobial therapy is prescribed after the isolation of acinetobacters from pathological material, the choice of antibiotic should be based on the antibiogram, taking into account the interpretive analysis of its results (section “Diagnostics and determination of sensitivity to antimicrobial drugs”).
Sulbactam. Sulbactam is currently the drug of choice for the treatment of Acinetobacter-associated infections. In the Republic of Belarus, 84.8% of hospital isolates are sensitive to this antimicrobial drug A. baumannii.
Sulbactam has intrinsic antimicrobial activity against A. baumannii, which does not depend on the beta-lactam drug in combination with it.
In experimental animal studies, the effectiveness of sulbactam was comparable to that of carbapenems against carbapenem-sensitive acinetobacters. In clinical trials, the sulbactam/beta-lactam combination showed similar efficacy compared to carbapenems in VAP and sepsis caused by multidrug-resistant isolates A. baumannii. Outcomes of treatment of sepsis caused by a multidrug-resistant strain A. baumannii, using sulbactam did not differ from the outcomes observed when treating sepsis caused by non-resistant antibiotics with other antibacterial drugs. A. baumannii .
When administered parenterally, the concentration of sulbactam in blood serum is 20-60 mg/l, in tissues - 2-16 mg/l. The optimal dosage regimen for sulbactam is 2 g as a 30-minute infusion every 6 hours or 1 g as a 3-hour infusion every 6-8 hours. When using high doses of sulbactam (3 g per administration), the development of adverse drug reactions in the form of diarrhea, rash, kidney damage.
As a result of a number of studies, a synergistic effect of sulbactam with meropenem, imipenem, rifampicin, cefpirome, and amikacin has been established.
Carbapenems. For the treatment of severe infections caused by A. baumannii, imipenem, meropenem and doripenem can be used. Ertapenem has no activity against Acinetobacterspp. generally .
Due to the increasing number of carbapenem-resistant strains A. bauma-nnii, including in the Republic of Belarus, the use of carbapenem antibiotics for the treatment of acinetobacter-associated infections in monotherapy is currently inappropriate. The exception is hospital healthcare organizations, where, according to local monitoring of antibiotic resistance of hospital pathogens, the absolute majority of the latter remain sensitive to carbapenems.
In research in vitro a synergistic or additive effect of the combinations imipenem + amikacin + colistin, doripenem + amikacin, doripenem + colistin, meropenem + sulbactam, meropenem + colistin has been established; in vivo- imipenem + tobramycin.
The use of a combination of carbapenem + beta-lactam/sulbactam for the treatment of bloodstream infections caused by multidrug-resistant A. baumannii, is associated with better treatment outcomes than the use of carbapenem monotherapy or the combination of carbapenem + amikacin. However, the combination of imipenem with sulbactam was associated with lower survival rates in a mouse model of pneumonia compared with the combination of imipenem + rifampicin.
When choosing a drug from this class for the treatment of Acinetobacter-associated infections, it is necessary to take into account that in the Republic of Belarus, imipenem has slightly greater activity against nosocomial isolates A. baumannii compared to meropenem (44.1 and 38.6% of susceptible strains, respectively). The activity of doripenem exceeds the activity of imipenem and meropenem only against isolates A. baumannii having the OXA-58 gene, the activity of imipenem is against OXA-23-producing strains A. baumannii. However, in the Republic of Belarus, OXA-40-producing strains of acinetobacteria predominate, which does not allow us to talk about the advantages of this drug over other representatives of the class in the treatment of infections caused by A. baumannii.
Aminoglycosides. Aminoglycosides are often used in the treatment of infections caused by gram-negative organisms, but hospital isolates A. baumannii have a high level of resistance to this class of antibacterial drugs. In the Republic of Belarus, 64.4% of the studied strains are resistant to gentamicin, and 89% of the studied strains are resistant to amikacin. A. baumannii. The relatively low level of resistance to gentamicin is most likely due to a decrease in the use of this antimicrobial drug in healthcare organizations over the past few years.
Prescription of this class of drugs is possible only in combination with antibiotics that are more active against acinetobacters based on local data on the sensitivity of the pathogen.
Rifampicin. Considering the sensitivity of hospital strains of acinetobacter to rifampicin, this drug can be added to the treatment of infections caused by multidrug-resistant strains. A number of authors have shown the effectiveness of rifampicin in monotherapy, as well as in combination with imipenem or sulbactam. Synergism is also characteristic of the combination of rifampicin with colistin. The effectiveness of rifampicin and the combination of rifampicin with colistin in meningitis caused by imipenem-resistant isolate has been shown A. baumannii .
According to a number of studies, resistance to rifampicin develops during treatment, both when used as monotherapy and in combination with imipenem, however, when using the combination of rifampicin + colistin, no changes in the MIC of rifampicin were shown.
Tetracyclines. Tetracyclines (minocycline, doxycycline, tetracycline) in studies invitro have activity against A. baumannii. The most active is minocycline (not registered in the Republic of Belarus), which is also active against isolates resistant to other tetracyclines. In general, experimental and clinical data characterizing the use of tetracyclines in infections caused by A. baumannii, are extremely few in number. Therefore, the prescription of drugs of this class is justified only on the basis of antibiogram data in the absence of another alternative.
Polymyxins. Of the five known drugs of this class (polymyxins A-E), only polymyxin B and polymyxin E (colistin) are currently available for clinical use. Colistin is used in two forms: colistin sulfate (for intestinal decontamination and for topical use for soft tissue infections; rarely for intravenous administration) and colistimethate sodium (for parenteral and inhalation administration). Colistimethate sodium (an inactive precursor to colistin) has less toxicity and antibacterial activity compared to colistin sulfate.
Polymyxins have high activity against strains A. baumannii, including multidrug - resistant and carbapenem - resistant isolates . According to various studies, the level of clinical effectiveness of colistin is 20-83%, microbiological 50-92%. According to pharmacokinetic studies, the concentration of colistin in the blood plasma after intravenous administration is in the range of 1-6 mg/l, in the cerebrospinal fluid - 25% of the serum concentration.
Due to poor penetration of histogematic barriers in patients with lower respiratory tract infections, polymyxins are preferably administered by inhalation, and in the treatment of infections of the central nervous system - intraventricularly or intrathecally, in combination with their parenteral administration or systemic use of other antimicrobial drugs.
The incidence of nephrotoxicity with the use of polymyxins, according to modern studies, is comparable to other classes of antibacterial drugs and amounts to 0-37%. The risk of developing nephrotoxicity when using polymyxins is dose dependent. At the same time, the highest incidence of side effects from the kidneys was observed in patients with previous impairment of their function, but the developing renal failure was usually reversible.
According to research in vitro there is synergy between colistin and rifampicin, imipenem, minocycline and ceftazidime; polymyxin B with imipenem, meropenem and rifampicin.
Currently, parenteral forms of polymyxins are not registered for use in the Republic of Belarus.
Tigecycline. Tigecycline has a bacteriostatic or bactericidal effect on A. baumannii, is not subject to resistance mechanisms characteristic of tetracyclines.
According to the results of a number of studies, tigecycline can maintain activity against minocycline-resistant, imipenem-resistant, colistin-resistant, multi-resistant strains A. baumannii .
Tigecycline has a large volume of distribution and creates high concentrations in body tissues, including the lung, however, according to some authors, the concentration of the drug in the blood and cerebrospinal fluid with the recommended administration regimen is suboptimal and does not provide sufficient antibacterial activity. Due to low concentrations of the drug in urine, the use of tigecycline for UTI is not recommended.
According to experts from the Food and Drug Administration (USA), tigecycline has been proven effective for the treatment of severe intra-abdominal infections caused by MSSA and VSE, severe skin and soft tissue infections caused by MSSA and MRSA, and community-acquired pneumonia. At the same time, the use of tigecycline for the treatment of nosocomial pneumonia (especially VAP) is associated with an increased risk of death in severe patients. The drug is not currently registered in the Republic of Belarus.
Table 4. Doses of antibacterial drugs and frequency of their administration
during treatment A. baumannii-associated infections
|
Preparation |
Dose and frequency of administration |
|
Ampicillin/sulbactam |
IV 12 g/day in 3-4 injections |
|
Cefoperazone/sulbactam |
IV 8.0 g/day in 2 injections |
|
Imipenem |
IV drip for 30 minutes in 100 ml of 0.9% sodium chloride solution, 1.0 g every 6-8 hours |
|
Meropenem |
IV drip for 15-30 minutes in 100 ml of 0.9% sodium chloride solution, 2.0 g every 8 hours |
|
Doripenem |
IV 1.5 g/day in 3 injections |
|
Netilmicin |
IV 4-6.5 mg/kg/day in 1-2 injections |
|
Amikacin |
IV 15-20 mg/kg/day in 1-2 injections |
|
Tobramycin |
IV 3-5 mg/kg/day in 1-2 injections |
|
Rifampicin |
IV 0.5 g/day in 2-4 doses |
|
Tigecycline* |
IV at a loading dose of 0.1 g, followed by 50 mg every 12 hours |
|
Colistin (colistimethate sodium*) |
IV 2.5-5 mg/kg/day in 2-4 injections; inhalation 1-3 million units every 12 hours |
* The drug is not registered in the Republic of Belarus.
Prospects for the treatment of infections caused by A. baumannii. In research in vitro Has the effectiveness of a new cephalosporin, ceftobiprole, been described? against Acinetobacterspp., however, there are no clinical trial data. The activity of ceftobiprole is superior to that of ceftazidime and cefepime in the absence or low expression of genes responsible for the synthesis of ADC-beta-lactamases. British authors in the study invitro showed the activity of the new monobactam BAL30072 against 73% CRAB at a concentration of 1 mg/L and 89% at 8 mg/L.
In the study invivo modeling of burn lesions in mice demonstrated the effectiveness of photodynamic therapy for the treatment of localized infections caused by multidrug-resistant A. baumannii .
Among the fundamentally new drugs being developed with potential activity against A. baumannii possess efflux pump inhibitors, inhibitors of bacterial fatty acid biosynthesis enzymes (FabI- and FabK-inhibitors), inhibitors of peptide deformylase metalloenzymes, antimicrobial peptides (buforin II, A3-APO), class D beta-lactamase inhibitors based on boronic acid. In the study invitro demonstrated the ability of the experimental drug NAB741, containing a cyclic polypeptide fragment identical to a similar region of polymyxin B, to increase sensitivity Acinetobacterbaumannii to drugs for which the intact outer membrane is an effective barrier. In another invitro The study showed the effectiveness of vancomycin against A. baumannii using fusogenic liposome technology to deliver it to the periplasmic space. The ability of substances that destroy biofilm (in particular, based on 2-aminoimidazole) to restore the sensitivity of multiresistant isolates of acinetobacter to antibiotics has been described. The possibility of developing so-called “antigens” aimed at inhibiting genes responsible for the formation of resistance mechanisms is being discussed; active and passive immunization. A number of studies have shown the activity of extracts from plants and animal secretions against multi-resistant acinetobacters. In particular, oil Helichrysumitalicum, tannic and ellagic acids significantly reduce the level of resistance A. baumannii to antibacterial drugs due to inhibition of efflux.
A number of studies have shown the lysis of Acinetobacter invitro, as well as the effectiveness of the use of bacteriophages in the treatment of experimental infections caused by Acinetobacter spp.., in animals.
Prevention
Given the high resistance Acinetobacterbaumannii to antimicrobial drugs, as well as the ability of this microorganism to quickly develop resistance mechanisms, prevention becomes of great importance A. baumannii- associated infections in a healthcare organization, which is based on the principles and standards of infection control.
A. baumannii capable of colonizing normally sterile objects and surviving in both dry and wet conditions of the hospital environment. Objects surrounding the patient (feathers in pillows, mattresses, bedding, curtains, beds, bedside tables and bedside tables, oxygen and water taps, water used in ventilators or for nasogastric administration), as well as those used to care for him, are usually subject to colonization , monitoring his condition, carrying out therapeutic manipulations. Among the items used for care and therapeutic procedures A. baumannii is isolated from artificial lung ventilation devices and mechanical suctions; objects associated with intravascular access (infusion pumps, pressure meters, long-term hemofiltration systems, vascular catheters) can also be colonized. Among other equipment, gurneys for transporting patients, medical gloves, gowns, blood pressure cuffs, peak flow meters, pulse oximeters, laryngoscope blades, and ventilation and air conditioning systems may be colonized. Thanks to the ability to exist in a humid environment A. baumannii contaminate a wide variety of solutions, including some disinfectants (furacilin, rivanol). Objects in the hospital environment that frequently come into contact with the hands of staff (door handles, computer keyboards, medical records, tables at medical stations, sinks and even cleaning equipment), floor coverings also serve as an additional reservoir A. baumannii .
During nosocomial outbreaks of infections caused by A. baumannii, medical procedures can also be associated with the spread of the pathogen, mainly due to contamination of the materials used. Such manipulations can be hydrotherapy or pulse lavage of wounds, surgical interventions, catheterization, tracheostomy, spinal puncture.
For adequate implementation of infection control of nosocomial A. baumannii-associated infections, it is necessary to constantly maintain measures aimed at preventing the transmission of the pathogen from patient to patient (Fig. 2), since the main reservoir A. baumannii inpatients are colonized/infected patients.

With the exception of the above measures, it is important to introduce strict indications for the prescription of antimicrobial drugs not included in the first line of antimicrobial therapy (for example, carbapenems, cephalosporins and fourth-generation fluoroquinolones, etc.), which reduces the frequency of inappropriate prescription of antibiotics in the hospital healthcare organization as a whole and , as a consequence, the resistance levels of hospital isolates, including A. baumannii.
In general, it should be said that Acinetobacter baumannii, is currently a “problematic” causative agent of nosocomial infections, affecting mainly patients in severe clinical condition, well adapted to living in a hospital environment and highly resistant to most antiseptic and antimicrobial drugs. When prescribing antibacterial therapy aimed at A. baumannii, it is necessary to take into account local data on its sensitivity in a specific healthcare organization, and more preferably in each specific department.
Medical news. - 2011. - No. 5. - pp. 31-39.
Attention!The article is addressed to medical specialists. Reprinting this article or its fragments on the Internet without a hyperlink to the source is considered a violation of copyright.
Hello,
The child has been sick since October of this year: first snot, a few days later a temperature of 39 and a cough (the temperature was brought down, the cough remained), then a purulent sore throat, then an increase in cough without a sore throat, then again a purulent sore throat with a temperature of 40 that could not be broken for three days. They gave injections of Cyfozalin — with a sore throat, the issue was closed. They began to treat the cough closely. They took tests (blood, urine, nasal and throat swabs). From the throat analysis: Acinetobacter baumannii complex (10 * 7 CFU, Klebsiella pneumoniae 10 * 7 CFU, Viridans Streptococcus Group 10 * 7 CFU). These smears were taken at a paid clinic. Based on the results of the analysis, we were told to contact an infectious disease specialist. We applied for a referral to an infectious disease specialist within the framework of compulsory medical insurance, but they refused, citing the fact that such tests are not prescribed now, since these infections go away on their own and do not need to be treated. What should we do?
Good day!
Today we received the results of a smear from the throat of a 4-year-old child, where this nasty thing was detected: Acinetobacter baumanii 10*5.
The ENT specialist somewhat surprised me, saying that she didn’t even know such a “beast,” but after examining it, without a second smear, she said that we had already defeated it (after a week’s treatment with candida). Naturally, I go to Google, there is not much information about this bacterium, at least in Russian. But from what I read I was already upset.
Please, anyone who has already encountered this, please share the opinions of your doctors: how dangerous is it and is it still treatable?
Microorganisms of the genus Acinetobacter are pleomorphic gram-negative rods that are easily confused with microorganisms of the genus Neisseria. Severe infections caused by these microorganisms, including meningitis, bacterial endocarditis, pneumonia, and bacteremia, are being described with increasing frequency.
Etiology
The A. calcoaceticus variant Iwoffi was described by DeBord as Mima polymorpha in 1939. It is one of two well-known variants of Acinetobacter. The other is the A. calcoaceticus variant of anitratus, first called Herella vaginicola. Microorganisms described as Bacterium anitratum and B5W may be designated as synonym Acinetobacter. These microorganisms are pleomorphic, encapsulated, nonmotile, and gram-negative. They grow well on simple nutrient media, forming white, convex, smooth colonies. Among microbes grown on solid media, diplococcal forms predominate; rod-shaped and filamentous variants of the pathogen are more often detected in liquid media. Species identification from Enterobacteriaceae is based on their negative reaction with nitrates, and from representatives of the genus Neisseria, which they may resemble in morphology, based on their unpretentiousness to nutrient media, their rod-shaped shape in liquid media, and their inherent negative oxidase reaction.
Epidemiology and pathogenesis
Microorganisms of the genus Acinetobacter are ubiquitous. They are normal inhabitants of the skin of 25% of healthy people. In cases of possible participation of Acinetobacter in bacteremias that develop during the use of intravenous catheters in a patient, the skin is considered the main entry point for infection. The increased incidence of Acinetobacter pneumonia, both primary and secondary to superinfection, also suggests that the respiratory tract may serve as an important portal of infection. In all likelihood, this microorganism is a common commensal with relatively low virulence, which causes bacterial carriage much more often than causes the development of infection. It appears that Acinetobacter diseases develop in individuals who unexpectedly encounter nosocomial Gram-negative infections. Severe infections develop in conditions of reduced resistance of the macroorganism, during instrumental interventions or after long-term antimicrobial therapy with broad-spectrum drugs. An unexplained increase in the incidence of Acinetobacter pulmonary infections in late summer has been noted. The participation of this microorganism in the etiology of conjunctivitis, vaginitis and urethritis requires further confirmation.
Manifestations
Severe infections caused by Acinetobacter include meningitis, subacute and acute bacterial endocarditis, pneumonia, urinary tract infections and bacteremia. Typically, the clinical manifestations and symptoms of this infection do not differ from those of similar diseases caused by other pathogens. Sometimes Acinetobacter can cause fulminant bacteremia with severe fever, vascular collapse, petechiae, massive subcutaneous hemorrhages, which are indistinguishable from meningococcemia. More often, however, bacteremia is associated with obvious introduction of infection through the venous system, in particular through venous catheters, surgical wounds or burn surfaces. It can also develop after instrumental interventions on the urethra or in other areas. The clinical manifestations of such diseases are dominated by endotoxemia, and their prognosis is unfavorable.
Diagnostics
Diagnosis of Acinetobacter infection can be difficult because clinical bacteriology laboratory personnel have little knowledge of these organisms, leading to misinterpretation of test results. The confusion observed in the taxonomic classification of these microorganisms also does not make the task easier. As a practical matter, isolation of Acinetobacter from blood, cerebrospinal fluid, sputum, urine or pus should be considered to be of clinical significance unless there is evidence of the co-occurrence of any other pathogen. In practical terms, differentiation of Acinetobacter and Neisseria is mandatory, since the former are resistant to penicillin, and the latter are sensitive.
Treatment
The sensitivity of individual Acinetobacter strains to antibiotics varies, but most are inhibited by gentamicin, tobramycin, amikacin, and ureidopenicillins such as piperacillin. Sensitivity to tetracyclines is unpredictable, and most strains are resistant to penicillin, ampicillin, cephalosporins, erythromycin and chloramphenicol. For severe systemic infections, an appropriate antibiotic, usually an aminoglycoside, should be used. Localized abscesses caused by these microorganisms require surgical drainage.
Acinetobacter is a ubiquitous, free-living, saprophytic microorganism that lives in healthy people on the skin, intestines and urogenital tract. These microbes usually colonize areas of the skin on the legs and groin. Most often, bacteria cause indolent diseases, as well as foodborne illnesses and traveler's diarrhea. Most microbes of the genus Acinetobacter are extremely resistant to therapy.
In 1911, Dutch microbiologist Martin Willem Beijerinck first isolated coccoid-shaped bacteria from soil that were capable of utilizing calcium acetate as an energy source. He named them Micrococcus calcoaceticus. A few years later, the properties of these microbes were described: immobility, inability to oxidize carbohydrates and reduce nitrates. Many authors have described such microorganisms. In 1954, the genus Acinetobacter was officially discovered, which included only oxidase-negative microorganisms. The term “acinetobacter” translated from ancient Greek means “immobile bacterium”, devoid of flagellar organelles of movement - flagella.
Acinetobacter spp. Until recently, they were considered low pathogenic microorganisms. Modern scientists have proven that under certain conditions the virulence of microbes increases. This leads to the development of severe infectious processes - and septicemia. Since 2017, Acinetobacter began to officially belong to the class of dangerous bacteria, which is due to their resistance to modern antimicrobial agents.
Acinetobacter spp. found in various natural objects: water, soil, wastewater. A. baumannii is a clinically significant member of this genus. It is a typical causative agent of nosocomial infections. In severely ill patients, A. baumannii causes sepsis, urethritis, and wound infection.
The growth of multidrug-resistant strains of Acinetobacter is a serious problem in modern medicine. Currently, bacteria have emerged that are resistant to all major groups of antibiotics. They are difficult to eliminate from the body. Medical scientists are actively searching for preventive measures and developing new medications that are active against such pathogens.
Etiology
Acinetobacter is a genus of non-fermenting microorganisms from the family Moraxellaceae.

Epidemiology
Acinetobacters readily inhabit any biotopes with minimally suitable conditions and contaminate a wide variety of objects. Strains of bacteria are found in all soil and water samples, on the skin and mucous membranes of the upper respiratory tract of healthy people.
Bacteria of the genus Acinetobacter are also isolated from pasteurized milk, frozen foods, hospital air, and swabs from various medical equipment. They have low virulence and are normal inhabitants of the human body. The appearance of microbes in large numbers on the skin, in sputum or urine does not indicate the development of an infectious process, but rather colonization or contamination.

The source and reservoir of infection are infected and sick people, as well as contaminated objects. The spread of bacteria is carried out by airborne droplets, household contact, and hematogenous routes.
Factors predisposing to the development of infection:
- Emergency hospitalization
- Severe concomitant diseases - hematological, oncological, endocrine,
- Long-term mechanical ventilation
- Inhalation administration of drugs,
- Invasive medical procedures - insertion of catheters and probes, tracheostomy,
- Premature and newborn babies in the first week of life,
- Treatment with cytostatics or hormones,
- Previous operations and transplantations,
- Long inpatient treatment
- Male gender and older age.
Acinetobacter infection is characterized by seasonal outbreaks in the summer season, which is associated with increased microbial colonization of the skin due to sweating.
A. baumannii is a microbe that lives primarily in water bodies: artificial and natural reservoirs. On a dry surface, the bacilli remain viable for a month. In health care institutions, Acinetobacter colonizes reusable solutions.
Acinetobacter spp. cause nasocomial infections in debilitated, physically weakened or mentally retarded patients. Microbes have an affinity for the epithelium of the respiratory and urogenital tracts, cerebrospinal fluid, blood, and peritoneal fluid. Immunocompromised patients usually develop pneumonia, bacteremia, septicemia, meningitis, endocarditis, brain and lung abscesses, pleural empyema, mediastenitis, and peritonitis.
Under stationary conditions, A. baumannii colonizes:
- Bedding, linen, furniture, water taps,
- Medical equipment - ventilators, infusion pumps, tonometers, thermometers, blood transfusion systems, catheters,
- Gloves, masks, gowns and other medical supplies,
- Various solutions,
- Materials for medical procedures - hydrotherapy, operations, catheterization, tracheostomy, lumbar puncture.
Symptoms
Acinetobacter is an opportunistic microorganism that causes an infectious process only when the immune defense is reduced.
Acinetobacter causes:
- Inflammation of the respiratory organs - paranasal sinuses, trachea and bronchi, lungs,
- Blood infection - bacteremia, septicemia,
- Pathology of the urogenital tract - cystitis, urethritis,
- Damage to skin and soft tissues,
- Diseases of the central nervous system - inflammation of the meninges and brain abscesses,
- Pathology of the intra-abdominal space - abscesses, peritonitis,
- Diseases of bones and joints - osteomyelitis, arthritis,
- Eye damage – unfavorable endophthalmitis and keratitis.
A. calcoaceticus is a causative agent of inflammatory processes in the lung tissue, urogenital tract, and blood. A. junii causes bacteremia and sepsis in patients, purulent inflammation of the subcutaneous fat. A. lwoffii and A. pittii are the causative agents of gastritis and colitis, and A. haemolyticus is the causative agent of bloody diarrhea.
 Bloodstream infections caused by A. baumannii occur in the form of bacteremia or sepsis. The entry gates are intravascular catheters. Microbes can enter the blood from existing sites - the urinary tract, infected soft tissues, burn surfaces, abdominal organs and the central nervous system. Fulminant bacteremia is manifested by severe fever, vascular collapse, petechiae, and massive subcutaneous hemorrhages. In the absence of effective therapy, 30% of patients develop infectious-toxic shock.
Bloodstream infections caused by A. baumannii occur in the form of bacteremia or sepsis. The entry gates are intravascular catheters. Microbes can enter the blood from existing sites - the urinary tract, infected soft tissues, burn surfaces, abdominal organs and the central nervous system. Fulminant bacteremia is manifested by severe fever, vascular collapse, petechiae, and massive subcutaneous hemorrhages. In the absence of effective therapy, 30% of patients develop infectious-toxic shock.
When the respiratory tract becomes infected, nosocomial pneumonia develops, which is characterized by simultaneous damage to several segments, the formation of cavities, pleural effusion, and the formation of a bronchopleural fistula. In patients with severe intoxication, a suffocating cough with purulent sputum appears, shortness of breath, breathing becomes noisy with moist rales. Acinetobacter pneumonia is severe and difficult to treat. It often ends in the death of patients.
A. baumannii is an important pathogen infecting burn surfaces and postoperative wounds. Skin and soft tissue infections are often complicated by bacteremia. This microbe is the causative agent of nasocomial meningitis and brain abscessation. Patients develop characteristic signs: intoxication, focal symptoms, meningeal signs. A petechial rash appears on the skin, pleocytosis appears in the cerebrospinal fluid, an increase in protein and lactic acid levels, and a decrease in glucose.
Acinetobacters in more rare cases cause:
- Peritonitis in persons on dialysis;
- Urinary tract infection during bladder catheterization and nephrolithiasis;
- Inflammation of the biliary tract after drainage;
- Osteomyelitis and arthritis caused by traumatic injury or installation of implants;
- Eye damage caused by infection of contact lenses.
Diagnostics
The main diagnostic method for acinetobacter infection is bacteriological. It consists in the correct collection of material, its rapid delivery to the laboratory, identification of the isolated pathogen, determination of its etiological significance and sensitivity to antibacterial agents.
The material is selected before the start of antimicrobial therapy directly from the source of infection in compliance with the rules of asepsis, preventing its contamination by foreign microflora. Selection is carried out with a sterile cotton swab, which is placed in special transport media. Liquid biomaterial is placed in sterile and tightly closed containers. Samples are delivered to the laboratory no later than 1.5–2 hours from the moment of collection.

The material is inoculated on liquid and solid nutrient media, incubated in a thermostat, and the results are taken into account. On dense media, smooth, opaque, shiny, small colonies are formed. On CA, after 48 hours, convex grayish-white colonies are formed, sometimes surrounded by a zone of hemolysis. Microscopic examination involves examining a stained specimen under a light microscope. Cocci and coccobacilli dominate in smears from native material, and rod-shaped forms dominate in smears from cultures. Acinetobacters are gram-negative microbes. After isolating a pure culture, the pathogen is identified by its biochemical properties. Acinetobacter does not ferment lactose and oxidizes glucose to acid.
Detection of acinetobacter in sputum in an amount of 106 CFU/ml is a diagnostically significant criterion. In bronchial lavage this amount is 104 CFU/ml, and in urine 105 CFU/ml.
Treatment
Treatment of Acinetobacter infections is a serious problem, the relevance of which is growing every day. This is due to an increase in the frequency of occurrence of microbes, an increase in their resistance to drugs and a decrease in the effectiveness of therapy.

Etiotropic antimicrobial treatment consists of the use of the following drugs:
- Imipenema or Meropenema
- "Amikatsina"
- "Ciprofloxacin"
- "Levofloxacin"
- "Ampicillin/sulbactam"
- "Cefoperazone/sulbactam"
- "Polymyxina"
- "Tetracycline"
- "Rifampicin"
- "Taigecycline".
If necessary, use combinations:
- "Cefoperazone/sulbactam" and "Amikacin"
- "Imipenem" and "Amikacin".
The choice of antibiotic is based on the results of the antibiogram.
Localized abscesses caused by these microorganisms require surgical drainage.
Acinetobacter spp. – a rather problematic causative agent of severe conditions and diseases encountered in pulmonological and therapeutic practice. Bacteria cause nosocomial pneumonia and cystic fibrosis, as well as a number of community-acquired pathologies. These microorganisms have natural resistance and acquired resistance. Most strains have multidrug resistance - resistance to the main groups of antibiotics. Members of the scientific medical society actively monitor the sensitivity status of such microbes and create formularies and standards for the use of antimicrobial agents.
Preventive measures
Specific prevention of Acinetobacter infection has not currently been developed. Nonspecific preventive measures are of great importance, since microorganisms are highly resistant to antibiotics. They quickly develop new resistance mechanisms.
Preventive measures for hospital infections are based on the principles and standards of infection control.
Measures to prevent infection with Acinetobacter:
- Carrying out disinfection and antiseptic procedures in medical institutions,
- Avoiding contact with sick people
- The use of personal protective equipment in crowded places during an epidemically dangerous period,
- Compliance with individual hygiene standards by medical staff,
- Rational prescription of antibiotics based on antibiogram data,
- Timely sanitation of foci of chronic infection - treatment of caries, tonsillitis, sinusitis,
- Strengthening the immune system - hardening, eating plant and lactic acid foods, playing sports, optimal work and rest regime, proper sleep, walks in the fresh air,
- Maintaining cleanliness of the body and home, regular ventilation of rooms, wet cleaning in public areas with chlorine-containing disinfectants,
- Scheduled medical examinations,
- Taking vitamin-mineral complexes in the spring and autumn.
Acinetobacter is a genus of microorganisms that predominantly cause nosocomial infections and affect seriously ill patients. These opportunistic microbes are normal inhabitants of various loci of the human body. Under certain conditions, they actively multiply and cause diseases of internal organs and systems that require specific antimicrobial treatment. The prognosis and outcome of acinetobacter infection depend on the pathogenicity of the strain, the activity of the host’s immune system, and the timeliness and accuracy of the prescribed treatment.
Video: Acinetobacter in the program “Live Healthy!”